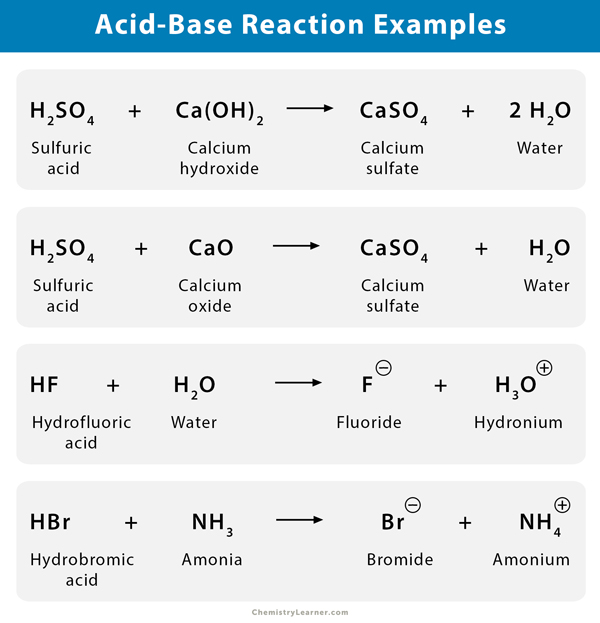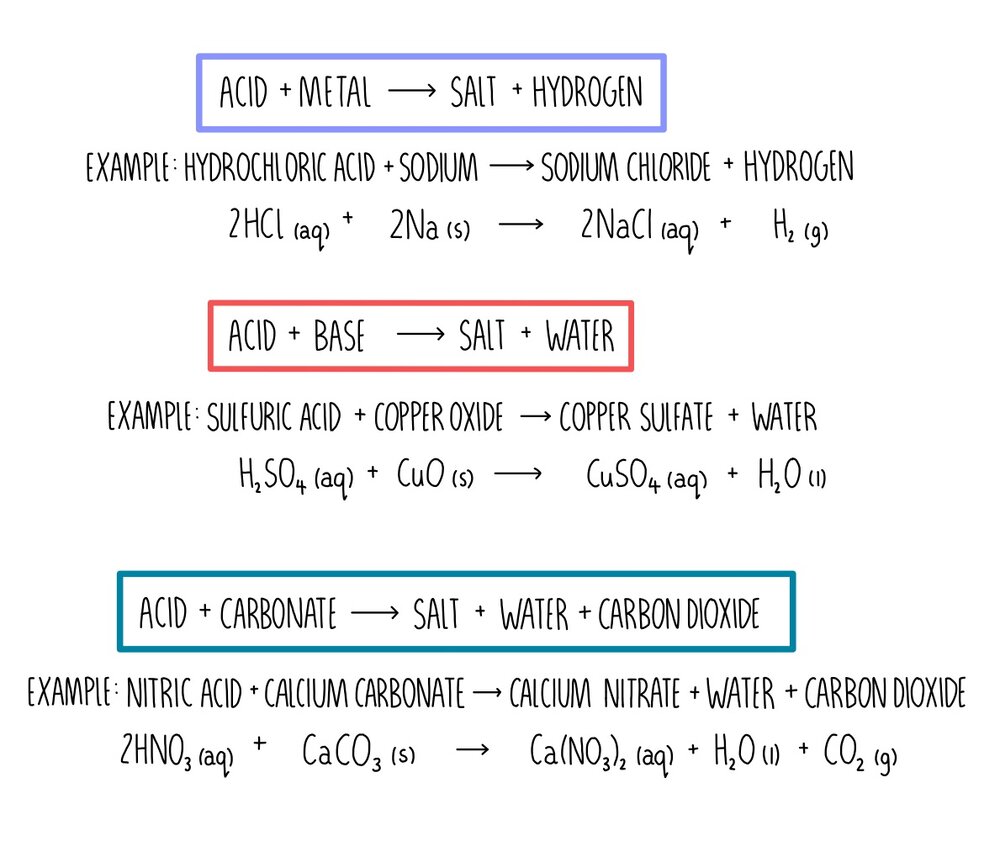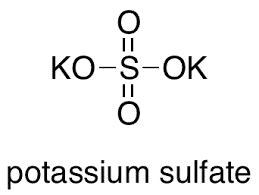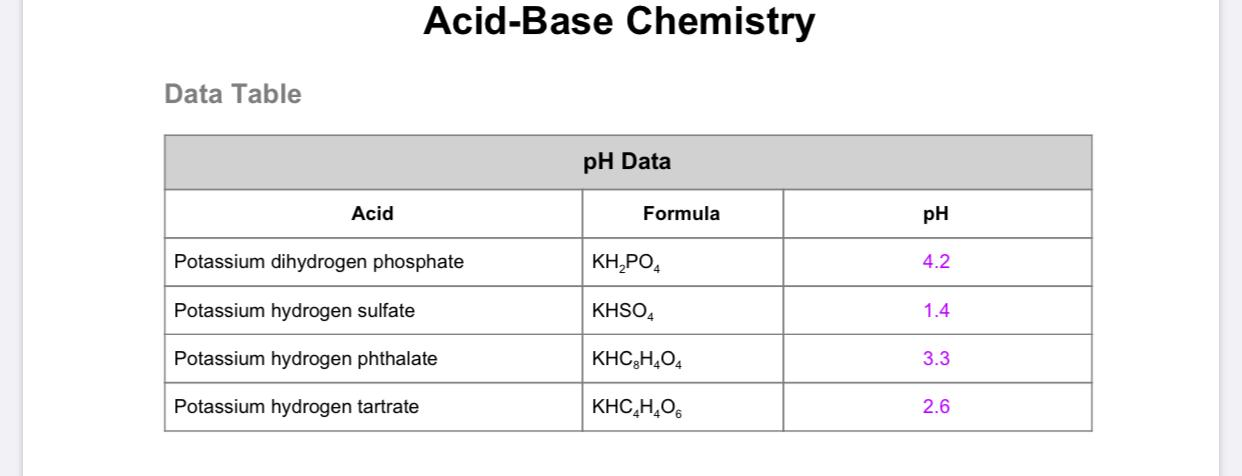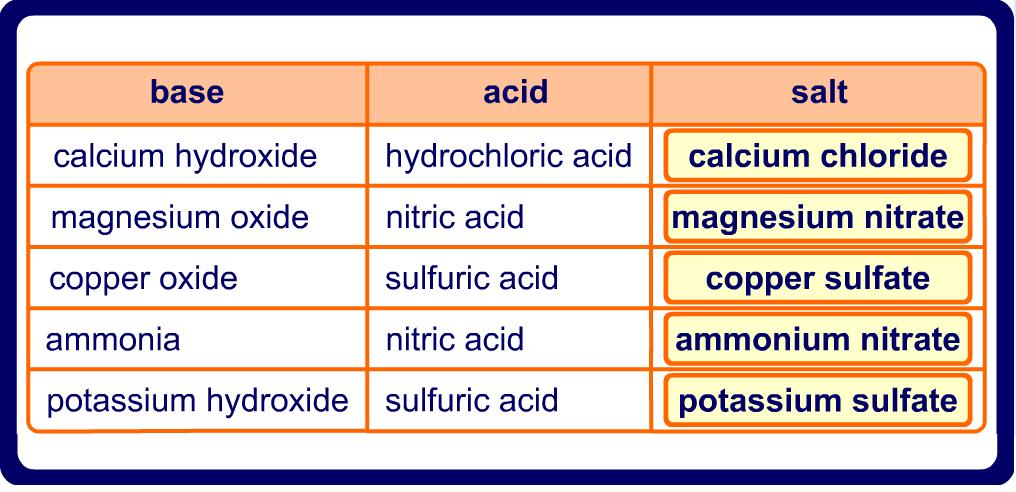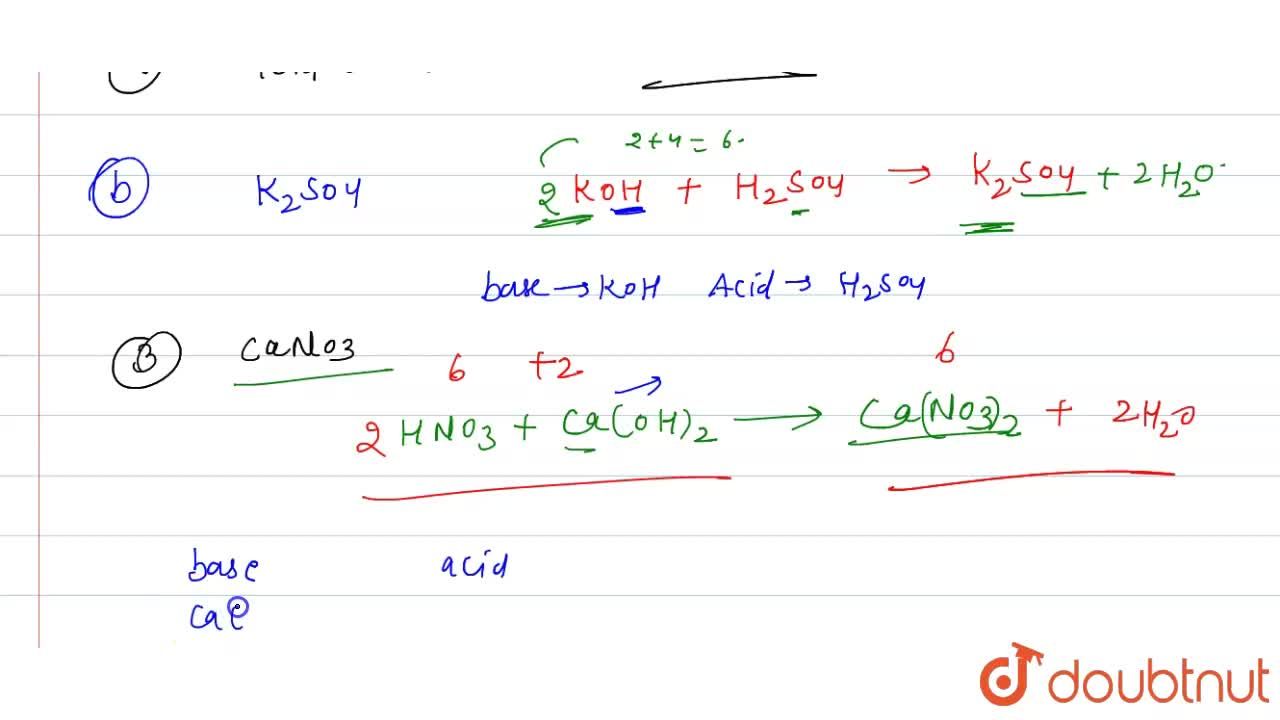
Name the acid and base from which the following salts are formed. (a) Sodium chloride (b) Potassium sulphate (c) Calcium nitrate

When sulfuric acid and potassium hydroxide neutralize each other to make water and potassium sulfate, how is water formed? | Socratic

Write the formulae of the salts given below:Potassium sulphate, sodium sulphate, calcium sulphate, magnesium sulphate, copper sulphate, sodium chloride, sodium nitrate, sodium carbonate and ammonium chloride.Identify the acids and bases from which
Write the formulae of the given salts as ' potassium sulphate, sodium sulphate, calcium sulphate, - CBSE Class 10 Science - Learn CBSE Forum